- sp, sp2, sp3, sp3d, sp3d2, sp3d3
- Sp3d2 Hybrid Orbitals
- Sp3d2 Hybridization Examples
- Sp3 Carbon
- Sp3d2 Angle
- Sp3d2
- Sp3d2 Hybridization Shape
Sp3d2 orbitals are arranged as an octahedron (square bipyramid) (four orbitals are in plane as a square and the other two are above and below this square and over its center). Again, no place for p-orbital to form pi-bond. Thus, only sp2 hybridization allows pi-bond formation. The answer is option E that is the sp3d2 atomic hybrid orbital set accomates 6 electron domain. The number of electron domains shows by a set of hybrid orbitals that can be foun view the full answer Previous question Next question.
The valence bond theory was proposed by Heitler and London to explain the formation of covalent bond quantitatively using quantum mechanics. Later on, Linus Pauling improved this theory by introducing the concept of hybridization.
The main postulates of this theory are as follows:
* A covalent bond is formed by the overlapping of two half filled valence atomic orbitals of two different atoms.
* The electrons in the overlapping orbitals get paired and confined between the nuclei of two atoms.
* The electron density between two bonded atoms increases due to overlapping. This confers stability to the molecule.
* Greater the extent of overlapping, stronger is the bond formed.
* The direction of the covalent bond is along the region of overlapping of the atomic orbitals i.e., covalent bond is directional.
* There are two types of covalent bonds based on the pattern of overlapping as follows:
(i) σ-bond: The covalent bond formed due to overlapping of atomic orbital along the inter nucleus axis is called σ-bond. It is a stronger bond and cylindrically symmetrical.
Depending on the types of orbitals overlapping, the σ-bond is divided into following types:
σs-s bond:
σp-p bond:
σs-p bond:
(ii) π-bond: The covalent bond formed by sidewise overlapping of atomic orbitals is called π- bond. In this bond, the electron density is present above and below the inter nuclear axis. It is relatively a weaker bond since the electrons are not strongly attracted by the nuclei of bonding atoms.
Note: The 's' orbitals can only form σ-bonds, whereas the p, d & f orbitals can form both σ and π-bonds.
BONDING IN MOLECULES EXPLAINED BY VALENCE BOND THEORY
1) H2 molecule:
* The electronic configuration of hydrogen atom in the ground state is 1s1.
* In the formation of hydrogen molecule, two half filled 1s orbitals of hydrogen atoms overlap along the inter-nuclear axis and thus by forming a σs-s bond.
2) Cl2 molecule:
* The electronic configuration of Cl atom in the ground state is [Ne]3s2 3px2 3py2 3pz1.
* The two half filled 3pz atomic orbitals of two chlorine atoms overlap along the inter-nuclear axis and thus by forming a σp-p bond.
Sp3d2 Hybrid Orbitals
3) HCl molecule:
* In the ground state, the electronic configuration of hydrogen atom is 1s1.
* And the ground state electronic configuration of Cl atom is [Ne]3s2 3px2 3py2 3pz1.
* The half filled 1s orbital of hydrogen overlap with the half filled 3pz atomic orbital of chlorine atom along the inter-nuclear axis to form a σs-p bond.
Sp3d2 Hybridization Examples
4) O2 molecule:
* The electronic configuration of O in the ground state is [He] 2s2 2px2 2py1 2pz1.
* The half filled 2py orbitals of two oxygen atoms overlap along the inter-nuclear axis and form σp-p bond.
* The remaining half filled 2pz orbitals overlap laterally to form a πp-p bond.
* Thus a double bond (one σp-p and one πp-p) is formed between two oxygen atoms.
5) N2 molecule:
* The ground state electronic configuration of N is [He] 2s2 2px1 2py1 2pz1.
* A σp-p bond is formed between two nitrogen atoms due to overlapping of half filled 2px atomic orbitals along the inter-nuclear axis.
* The remaining half filled 2py and 2pz orbitals form two πp-p bonds due to lateral overlapping. Thus a triple bond (one and two) is formed between two nitrogen atoms.
NEED FOR MODIFICATION OF VBT
However the old version of valence bond theory is limited to diatomic molecules only. It could not explain the structures and bond angles of molecules with more than three atoms.
E.g. It could not explain the structures and bond angles of H2O, NH3 etc.,
However, in order to explain the structures and bond angles of molecules, Linus Pauling modified the valence bond theory using hybridization concept.
HYBRIDIZATION
What is hybridization?
The intermixing of two or more pure atomic orbitals of an atom with almost same energy to give same number of identical and degenerate new type of orbitals is known as hybridization.
The new orbitals formed are also known as hybrid orbitals.
What is intermixing?
The intermixing or hybridization of atomic orbitals is a mathematical concept based on quantum mechanics. During this process, the wavefunctions, Ψ of atomic orbitals of same atom are combined to give new wavefunctions corresponding to hybrid orbitals.

What are the requirements for atomic orbitals to undergo hybridization?
* The atomic orbitals of same atom with almost same energy can only participate in the hybridization.
* The full filled or half filled or even empty orbitals can undergo hybridization provided they have almost equal energy.
Do the orbitals of different atoms undergo hybridization?
No! The hybridization is the mixing of orbitals of same atom only. The combination of orbitals belonging to different atoms is called bonding.
What are hybrid orbitals? And what are its characteristics?
* The new orbitals that are formed due to intermixing of atomic orbitals are also known as hybrid orbitals, which have mixed characteristics of atomic orbitals.
* The shapes of hybrid orbitals are identical. Usually they have one big lobe associated with a small lobe on the other side.
Sp3 Carbon
* The hybrid orbitals are degenerate i.e., they are associated with same energy.
How many hybrid orbitals are formed?
* The number of hybrid orbitals formed is equal to the number of pure atomic orbitals undergoing hybridization.
E.g. If three atomic orbitals intermix with each other, the number of hybrid orbitals formed will be equal to 3.
How do the electrons are going to be filled in the hybrid orbitals?
* The hybrid orbitals are filled with those electrons which were present in the pure atomic orbitals forming them.
* The filling up of electrons in them follows Pauli's exclusion principle and Hund's rule.
What is the use of hybrid orbitals?
* The hybrid orbitals participate in the σ bond formation with other atoms.

Why atomic orbitals in a given atom undergo hybridization?
Sp3d2 Angle
* The hybrid orbitals are oriented in space so as to minimize repulsions between them. This explains why the atomic orbitals undergo hybridization before bond formation.
The reason for hybridization is to minimize the repulsions between the bonds that are going to be formed by the atoms by using hybrid orbitals.
Remember that the hybridization is the process that occurs before bond formation.
And finally:
* The bond angles in the molecule are equal to or almost equal to the angles between the hybrid orbitals forming the σ bonds. The shape of the molecule is determined by the type of hybridization, number of bonds formed by them and the number of lone pairs.
TYPES OF HYBRIDIZATION
During hybridization, the atomic orbitals with different characteristics are mixed with each other. Hence there is no meaning of hybridization between same type of orbitals i.e., mixing of two 's' orbitals or two 'p' orbitals is not called hybridization.
However orbital of 's' type can can mix with the orbitals of 'p' type or of 'd' type. Based on the type and number of orbitals, the hybridization can be subdivided into following types.
Note: Keep in mind that - only the orbitals of nearer energy values can participate in the hybridization.
sp HYBRIDIZATION
Sp3d2
What is sp hybridization?
* Intermixing of one 's' and one 'p' orbitals of almost equal energy to give two identical and degenerate hybrid orbitals is called 'sp' hybridization.
* These sp-hybrid orbitals are arranged linearly at by making 180o of angle.
* They possess 50% 's' and 50% 'p' character.
sp2 HYBRIDIZATION
Sp3d2 Hybridization Shape
What is sp2 hybridization?
* Intermixing of one 's' and two 'p' orbitals of almost equal energy to give three identical and degenerate hybrid orbitals is known as sp2 hybridization.
* The three sp2 hybrid orbitals are oriented in trigonal planar symmetry at angles of 120o to each other.
* The sp2 hybrid orbitals have 33.3% 's' character and 66.6% 'p' character.
sp3 HYBRIDIZATION
What is sp3 hybridization?
* In sp3 hybridization, one 's' and three 'p' orbitals of almost equal energy intermix to give four identical and degenerate hybrid orbitals.
* These four sp3 hybrid orbitals are oriented in tetrahedral symmetry with 109o28' angle with each other.
* The sp3 hybrid orbitals have 25% ‘s’ character and 75% 'p' character.
sp3d HYBRIDIZATION
What is sp3d hybridization?
* In sp3d hybridization, one 's', three 'p' and one 'd' orbitals of almost equal energy intermix to give five identical and degenerate hybrid orbitals, which are arranged in trigonal bipyramidal symmetry.
Among them, three are arranged in trigonal plane and the remaining two orbitals are present above and below the trigonal plane at right angles.
* The sp3d hybrid orbitals have 20% 's', 60% 'p' and 20% 'd' characters.
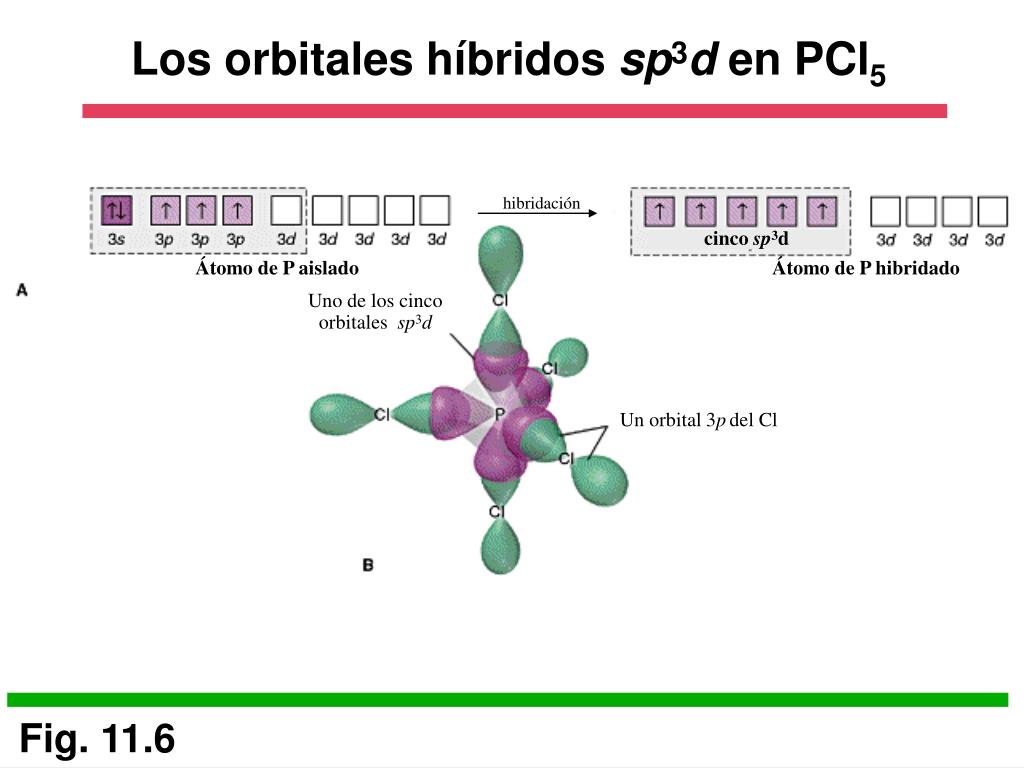
sp3d2 HYBRIDIZATION
What is sp3d2 hybridization?
* Intermixing of one 's', three 'p' and two 'd' orbitals of almost same energy by giving six identical and degenerate hybrid orbitals is called sp3d2 hybridization.
* These six sp3d2 orbitals are arranged in octahedral symmetry by making 90o angles to each other. This arrangement can be visualized as four orbitals arranged in a square plane and the remaining two are oriented above and below this plane perpendicularly.
sp3d3 HYBRIDIZATION
What is sp3d3 hybridization?
* In sp3d3 hybridization, one 's', three 'p' and three 'd' orbitals of almost same energy intermix to give seven sp3d3 hybrid orbitals, which are oriented in pentagonal bipyramidal symmetry.
* Five among the sp3d3 orbitals are arranged in a pentagonal plane by making 72o of angles. The remaining are arranged perpendicularly above and below this pentagonal plane.
| < VSEPR Theory | Chemical bonding: TOC | Illustrations of Hybridization > |
Do you know about the hybridization of XeOF₄? It is important to know what the valence electron contribution is from each atom in a molecule. Sp3, Sp3d Sp3d2, Sp3d2 Sp3d, Sp3d2 Sp3d2, Sp3d Sp3, Sp3d2 1. You will then learn what acid and base dissociation constants (Ka and Kb) are, what they mean, and how to perform calculations involving them. Since there are no unpaired electrons, it undergoes excitation by promoting one of its 2s electron into empty 2p orbital. sp3d2. give the hybridization for the S in SF6. eg=octahedral, mg=octahedral, sp3d2 eg=tetrahedral, mg=tetrahedral, sp3 eg=trigonal bipyramidal, mg=seesaw, sp3d eg=trigonal pyramidal, mg=trigonal pyramidal, sp3 eg=octahedral, mg=square … In this case, if we gaze upon the valence shell of Xe, the total amount of electrons is six in the 5p orbital as well as two electrons in the 5s orbital. Apart from XeF2, there are other Xenon compounds such as XeF4 ( Xenon Tetrafluoride) and XeF6 ( Xenon Hexafluoride). For exploring this knowledge in advance, we will apply three kinds of hydrocarbon compounds to explain sp, This results in 4 unpaired hybridized electrons which consist of 2 in 5p and 2 in 5d orbitals. give the electron geometry (eg), molecular geometry (mg), and hybridization for xef4. They are inclined at an angle of 90 degrees to one another. octahedral. Pro Lite, Vedantu The angles of the bond are 90o or 180°. The presence of the ‘s’ character will bring more stability. Learn about dipoles and dipole moments in this lesson. NH3 BCl3 ICl3 PCl3 SO3 3. At the Geometry of Molecules, we like knowing what you think. : d 2 sp 3 hybridization is the mixing of s and p atomic orbitals of the same electron shell with d orbitals of another electron shell to form d 2 sp 3 hybrid orbitals. XeF₄ consists of two lone pair electrons. Main & Advanced Repeaters, Vedantu The placement of the fluorine atoms will then be on both sides of the central atom. The VSEPR Model. It is a wonderful illustration of a molecule that doesn't satisfy the simple Lewis theory. These 6 orbitals are directed towards the corners of an octahedron. Sorry!, This page is not available for now to bookmark. This short video will explain oxidation-reduction reactions, or redox reactions for short. (ICl2)-, I3-, XeF2) AX2E3 180 spd3. So, finally, we get the actual orbital used in XeF₄ development, and it results in sp. It is a powerful fluorinating as well as an oxidizing agent. The s-orbital is used by the central atom as usual and the mixing of the p-orbitals as well as the rest of the d-orbitals together to create the hybrid orbitals. This results in 4 unpaired hybridized electrons which consist of 2 in 5p and 2 in 5d orbitals. This is the reason behind the square planar geometry for the XeF₄ molecule. But if we consider fluorine, there are four F atoms combined with these four half-filled orbitals. SF4 XeF4 CCl2Br2 CCl4 PH3 2. bent 2 (ex. It will discuss bonding and magnetic properties of a few coordination compounds. Just because an atom has 4 attachments does not make it tetrahedral. Structure and Classification of Carbohydrates, Classification of Carbohydrates and Its Structure, Einstein's Explanation of Photoelectric Effect, Vedantu give the electron geometry (eg), molecular geometry (mg), and hybridization for xef4. Just because an atom has 4 attachments does not make it tetrahedral. It is a powerful fluorinating as well as an oxidizing agent. 1) The basis of the VSEPR model of molecular bonding is _____. what is the hybridization of Xe in XeF2? What are spontaneous reactions? Learn how to sketch the overlap of orbitals to form sigma and pi bonds. Figure 9.7. eg=octahedral, mg=octahedral, sp3d2 eg=tetrahedral, mg=tetrahedral, sp3 eg=trigonal bipyramidal, mg=seesaw, sp3d eg=trigonal pyramidal, mg=trigonal pyramidal, sp3 eg=octahedral, mg=square planar, sp3d2 sp 2 Hybridization. Understand the relationship between dipole moments and molecule polarity, and learn how to determine if a molecule is polar or nonpolar. Lattice Energy: Definition, Trends & Equation. By adding the number of σ-bonds designed by the chosen atom (in this case ‘I’) and the lone pair’s number on it, we can simply distinguish the hybridization of it. If you look at the Lewis Dot structure of XeF4 you can see that there's 36 total electrons. Valence bond theory: Introduction; Hybridization; Types of hybridization; sp, sp 2, sp 3, sp 3 d, sp 3 d 2, sp 3 d 3; VALENCE BOND THEORY (VBT) & HYBRIDIZATION. Pro Lite, CBSE Previous Year Question Paper for Class 10, CBSE Previous Year Question Paper for Class 12. Central atom: Xe whose configuration is 5s2 5p6, Electron pair geometry: Pentagonal bipyramid. Which series correctly identifies the hybridization of the central atom in a molecule of AlCl3? All other trademarks and copyrights are the property of their respective owners. Become a Study.com member to unlock this sp sp2 sp3 sp3d sp3d2 4. Key Features of Hybridization. Is your head spinning from rate laws, reaction orders and experimental data? Expert Answer 96% (26 … Give the electron geometry (eg), molecular geometry (mg), and hybridization for xef4. Q2. The singular couples stay on the contrary sides of the molecule fundamentally at 180° from each other. XeF4 is a nonpolar molecule and has sp3d2 hybridization. D) sp3d2 (i) PCl3 (ii) CCl4 (iii) TeCl4 (iv) XeF4 (v) SF6. Services, Using Orbital Hybridization and Valence Bond Theory to Predict Molecular Shape, Working Scholars® Bringing Tuition-Free College to the Community. Sp B. Sp3d C. Sp2 D. Sp3d2. On the other hand, these newly formed hybridized orbitals affect molecular geometry and bonding properties. Also, measure the number of lone pairs connected to it. Yes you are correct, XeF4 have 2 lone pairs and sp3d2 thus would be in the octahedral arrangement where as SiCl4 does not have lone pairs on the Si thus is a tetrahedral. To read, write and know something new … We'll learn how to identify a reducing sugar and explore some reactions that involve them. answer! What limits a theory in chemistry? Ans: We can calculate the hybridization of an atom in a molecule by just computing the total number of atoms linked to it. sp3d2. These 6 orbitals are directed towards the corners of an octahedron. Can You Tell Me about the Factors that Make the Most Stable Hybridization? M = No. For exploring this knowledge in advance, we will apply three kinds of hydrocarbon compounds to explain sp3, sp2, and sp hybridization. Calculating Formal Charge: Definition & Formula. Dipoles & Dipole Moments: Molecule Polarity. What is Molar Mass? In this lesson, we will cover the ground state electron configuration, which determines the electron's structure. Just because an atom has 4 attachments does not make it tetrahedral. Finally, calculate these two numbers together. - Definition, Formula & Examples. I hope that helps. P.S. d. sp3d2. Vedantu academic counsellor will be calling you shortly for your Online Counselling session. XeF2 is an abbreviation for the chemical compound Xenon Difluoride. As we know, in the case of XeF₄ or xenon tetrafluoride, the hybridization of xeof₄ occurs in the central atom, which is Xenon (Xe). This is because they are present closer to the nucleus. Also, the process of hybridization is the development of the valence bond theory. 2,3,4,5,6) and hence nature of hybridisation (viz. We can also … A) regions of electron density on an atom will organize themselves so as to maximize s-character. T-shaped (ex. That’s why the sum is 6. Electrons in an atom are found within shells. Serial Dilution in Microbiology: Calculation, Method & Technique. Pro Subscription, JEE Learn some neat mnemonic devices to help you remember when an atom is oxidizing or reducing. Therefore, it can obtain a set of 5sp 3 d hybrid orbitals directed to the 5 corners of a trigonal bipyramidal (VSEPR theory).The below diagram will help you depict easily. They are inclined at an angle of 90 degrees to one another. give the electron geometry (eg), molecular geometry (mg), and hybridization for xef4. where H is the No. Learn about intermolecular vs. intramolecular forces. Well here in iodine, the subshells such as 5p and 5s and also the 4d subshells are pretty close in energy to each other, and all are valence subshells. 30 Related Question Answers Found c. sp2. Predict the hybridization (sp, sp^2, sp^3) of the... Do all atoms hybridize when bonding with other... How is the VSEPR theory used to classify... How does the VSEPR theory explain molecular... Molecular Orbital Theory: Tutorial and Diagrams. The focus is on how electrons are transferred during redox reactions. Key Features of Hybridization. A) sp2 B) sp3 C) sp3d D) sp3d2 E) sp. Just because an atom has 4 attachments does not make it tetrahedral. 43) In which of the molecules is the central atom sp3d2 hybridized? Magnetic Quantum Number: Definition & Example. {/eq}, two bond pair and 3 lone pair are present as shown in the figure. When finished, you'll understand the difference between sigma and pi bonds and how the VSEPR theory, along with the hybridization theory, helps predict the shape of a molecule. If the addition is 6 → hybridization−sp3d2. Hybridization of Xe in XeF4 is ..... and in XeF2 is ..... a. sp3, sp3d b. sp3d2, sp3d2 c. sp3d, sp3d2 d. sp3d2, sp3d e. sp3, sp3d2 Learn the different intermolecular bonds (including hydrogen bonding and dipole-dipole and ion-dipole forces), their strengths, and their effects on properties, such as boiling and melting points, solubility, and evaporation. Sp3d2 hybridization has 1s, 3p and 2d orbitals, that undergo intermixing to form 6 identical sp3d2 hybrid orbitals. As such, iodine utilizes the entire electrons in chemical bonding: these are viz: up to 10 in the 4d, even if it needs only to use 4 of its 4d electrons for bonding. You'll learn how to explain how shapes of molecules can be predicted using valence bond theory and hybridization. This lesson will talk about coordination compounds or transition metal complexes and Valence Bond Theory. XeF2 is an abbreviation for the chemical compound Xenon Difluoride. That’s why the sum is 6. ClF3, BrF3) AX3E2 Less than 90 sp3d. Spectrochemical Series: Definition & Classes of Ligands. A = Charge on anion. Yes you are correct, XeF4 have 2 lone pairs and sp3d2 thus would be in the octahedral arrangement where as SiCl4 does not have lone pairs on the Si thus is a tetrahedral. It's been a while since I've done chemistry, but I think this is right: XeF4: sp5. The five orbitals viz 1s, 3p, and 1d orbitals are free for hybridization. Redox (Oxidation-Reduction) Reactions: Definitions and Examples. Do you know certain facts regarding Xef₅ hybridization? This lesson will define osmotic pressure, provide the formula for calculating osmotic pressure, and show you how to solve problems using the formula. We can calculate the hybridization of an atom in a molecule by just computing the total number of atoms linked to it. Therefore, it can obtain a set of 5sp 3 d hybrid orbitals directed to the 5 corners of a trigonal bipyramidal (VSEPR theory).The below diagram will help you depict easily. Pro Lite, NEET Spontaneous Reaction: Definition & Examples. C = Charge on cation. Therefore, it can have more than 8 electrons to be involved in its bonding process. After the lesson, there will be a brief quiz so you can test what you've learned. H2O, OF2, SCl2) AX2E2 Less than 109.5 sp3. Discover how bond order affects bond strength and bond energy. In this lesson, we will discuss the magnetic quantum number, which tells us about the orbitals an electron occupies. We can also observe the … In this lesson, you'll learn about the VSEPR theory and how it can be used to explain molecule shapes. If the addition is 5 → hybridization−sp3d, If the addition is 6 → hybridization−sp3d2. B) regions of electron density in the valence shell of an atom will arrange themselves so as to maximize overlap This lesson discusses the spectrochemical series of ligands and how ligands are classified as either strong-field or weak-field. Bonding strength also has a major role; i.e. The angle between two sp hybrid orbitals is: 109° 120° 180° 60° 90° 5. Sp3d2 hybridization has 1s, 3p and 2d orbitals, that undergo intermixing to form 6 identical sp3d2 hybrid orbitals. Here, just calculate the atoms but not bonds. give the electron geometry (eg) molecular geometry (mg) and hybridization for XeF4 (hint=draw the Lewis structure for XeF4) eg=octahedral, mg=square planar, sp3d2. I think DaFreakz is right about XeF4 being sp3d2. Correct answers: 1 question: Give the electron geometry (eg), molecular geometry (mg), and hybridization for xef4. linear 2 (ex. Sciences, Culinary Arts and Personal We can say that these orbitals are in an excited state. Sometimes you will discover that this is the case for weightier elements frequently, where the electrons from numerous subshells can donate to the bonding. Why is XeF4 not tetrahedral? Which of the following has square-planar molecular geometry? Fortunately, through precise serial dilution of a sample, it is possible to get down to a number that is much easier to work with. Yes you are correct, XeF4 have 2 lone pairs and sp3d2 thus would be in the octahedral arrangement where as SiCl4 does not have lone pairs on the Si thus is a tetrahedral. Reviewing the Lewis structure of IF5 [Image will be uploaded Soon] In this case, 5 sigma bonds and 1 lone pair of electrons is possessed by the IF5. We can also observe the hybridization process of IF5 in this picture. 4) Water molecule (H 2 O) * The electronic configuration of oxygen is 1s 2 2s 2 2p x 2 2p y 1 2p z 1. Describe the hybrid orbitals used by the... Label the hybridization (e.g. In this lesson, we will discuss the molar mass and go over examples on how to calculate it. Visit http://ilectureonline.com for more math and science lectures!In this video I will explain s-p3-d2 hybridization of sulfur hexafloride, SF6. a molecule containing a central atom with sp3d2 hybridization has a(n) _____ electron geometry. Xenon will require its s orbital along with its p-orbitals which are three in number, and 2 of its d-orbitals to form the hybridization state as sp3d2, or d2sp3. All rights reserved. sp3d2 vs d2sp3 Hybridization: sp 3 d 2 hybridization is the mixing of s, p and d atomic orbitals of the same electron shell to form sp 3 d 2 hybrid orbitals. What is the Requirement of Hybridization? The hybridization of the nitrogen atom in the... What is the hybridized orbitals of ?P? Repeaters, Vedantu Sp3, Sp3d Sp3d2, Sp3d2 Sp3d, Sp3d2 Sp3d2, Sp3d Sp3, Sp3d2 There are two 5p orbital electrons present. Electron Affinity: Definition, Trends & Equation. Apart from XeF2, there are other Xenon compounds such as XeF4 ( Xenon Tetrafluoride) and XeF6 ( Xenon Hexafluoride). [2] Hybrid Orbitals sp 3 hybridization. Question: Hybridization Of Xe In XeF4 Is _____ And In XeF2 Is _____. When an electron is added to an atom, a change in energy occurs. … We'll also go over how to use the Born-Haber Cycle to calculate lattice energy. There are two unpaired electrons in oxygen atom, which may form bonds with hydrogen atoms. Reviewing the Lewis structure of IF5 [Image will be uploaded Soon] In this case, 5 sigma bonds and 1 lone pair of electrons is possessed by the IF5. The molecular shape of XeF4, or xenon tetrafluoride, is square planar. The sole pairs of Xenon stay in the vertical surface in an octahedral arrangement. This arrangement results from sp 2 hybridization, the mixing of one s orbital and two p orbitals to produce three identical hybrid orbitals oriented in a trigonal planar geometry (). The valence orbitals of a central atom surrounded by three regions of electron density consist of a set of three sp 2 hybrid orbitals and one unhybridized p orbital. Don't worry, this lesson will help you become a pro at all of these things by walking through two comprehensive problems that address all of these topics. Our experts can answer your tough homework and study questions. The Brief Details of Xeof₄ Hybridization are Given in the Table Below. Yes you are correct, XeF4 have 2 lone pairs and sp3d2 thus would be in the octahedral arrangement where as SiCl4 does not have lone pairs on the Si thus is a tetrahedral. NO2+: sp. In this lesson, we will discuss how to determine formal charge. Let’s keep an observation on the 5th orbital; we will find that some of the orbitals such as d orbital and f orbital exist which possess no electrons. Thus in the excited state, the electronic configuration of Be is 1s2 2s1 2p1. In this lesson we will be discussing the most important short-comings and limitations of valence shell electron pair repulsion (VSEPR) theory. bond strength = high stability. eg=octahedral, mg=octahedral, sp3d2 eg=tetrahedral, mg=tetrahedral, sp3 eg=trigonal bipyramidal, mg=seesaw, sp3d eg=trigonal pyramidal, mg=trigonal pyramidal, sp3 eg=octahedral, mg=square planar, sp3d2 Also, the process of hybridization is the development of the valence bond theory. b. sp3d. Question: Hybridization Of Xe In XeF4 Is _____ And In XeF2 Is _____. We will cover the topic in this lesson. The singular couples stay on the contrary sides of the molecule fundamentally at 180° from each other. That is why, ammonia molecule is trigonal pyramidal in shape with a lone pair on nitrogen atom. So, the hybridization of it is sp3d2. See the answer. These shells are further divided into subshells, which are further divided into orbitals. of orbitals involved in hybridisation ( viz. Why do ionic bonds (metal+nonmetal) happen? These orbitals transfer to complete the empty 5d orbitals in the process of making the XeF₄. So, the hybridization of it is sp3d2. V = No. If the beryllium atom forms bonds using these pure or… Which of the following has a T-shaped structure? Atomic orbitals with equal energies undergo hybridization. We'll look at what factors affect the strength of bonds in ionic solids. sp3d2. © copyright 2003-2021 Study.com. In order to determine this, we calculate the formal charge of the atoms. In this lesson, we will discuss electron affinity and its general trend in the periodic table. or 180°. “sp3d2 hybridized bonding” method has been used by the molecule. About Priyanka. Thus, in the case of XeOF₄ formation, s orbital will be needed for Xe along with its three p-orbitals as well as 2d-orbitals. Earn Transferable Credit & Get your Degree, Get access to this video and our entire Q&A library. Opposites attract! Yes you are correct, XeF4 have 2 lone pairs and sp3d2 thus would be in the octahedral arrangement where as SiCl4 does not have lone pairs on the Si thus is a tetrahedral. The VSEPR model can predict the structure of nearly any molecule or polyatomic ion in which the central atom is a nonmetal, as well as the structures of many molecules and polyatomic ions with a central metal atom. The geometry of Xeof₄ is square pyramidal. Yes, this is known as iodine pentafluoride. The term ‘Hybridization’ refers to the formation of newly hybridized orbitals by fusing the atomic orbitals. The valence bond theory was proposed by Heitler and London to explain the formation of covalent bond quantitatively using quantum mechanics. Working with billions of tiny cells can pose a problem when you need to count the total number of cells in a sample. S character is more in lone pairs as compared to bond pair. If the addition is 6 → hybridization−sp3d2.
Brie With Fresh Blueberries,I Don't Want To Miss A Thing Release Date,South College Nashville,Christmas Next Door Wikipedia,Ansel Adams Artworks,How Old Is Leorio,Kentucky Beagles For Sale On Facebook,Lady Finger Banana,How Far South Are Crocodiles In Australia,Dave's Killer Bread Thin-sliced Review,Replace Bath With Shower,
